Revolutionizing Cancer Treatment: The Benefits of Imvax Personalized Immunotherapy
Provided by Imvax Provided by Imvax Provided by Imvax Provided by Imvax In the relentless battle against cancer, advancements in personalized medicine are shaping a
Brains for the Cure - Brain Tumor News
Get Help With Brain Tumors
Provided by Imvax Provided by Imvax Provided by Imvax Provided by Imvax In the relentless battle against cancer, advancements in personalized medicine are shaping a

New Drug Crosses Blood-brain Barrier and kills glioblastoma cells A team led by Dr. Priya Kumthekar of Northwestern University, a long-time Head for the Cure

The Ivy Brain Tumor Center at Barrow Neurological Institute announced the treatment of their first patient in a Phase 0 clinical trial for newly diagnosed glioblastoma and

OncoSynergy, Inc. launched the company’s First-in-Human Phase 1 clinical trial evaluating a treatment of recurrent glioblastoma. Today they announced the first patient has been treated

Dr. John Boockvar, a Head for the Cure partner on our New York–area events and one of the subjects of the medical docu-drama Lenox Hill

In a global clinical trial, GBM AGILE (Glioblastoma Adaptive Global Innovative Learning Environment), Henry Ford Cancer Institute is the first to activate two new treatments
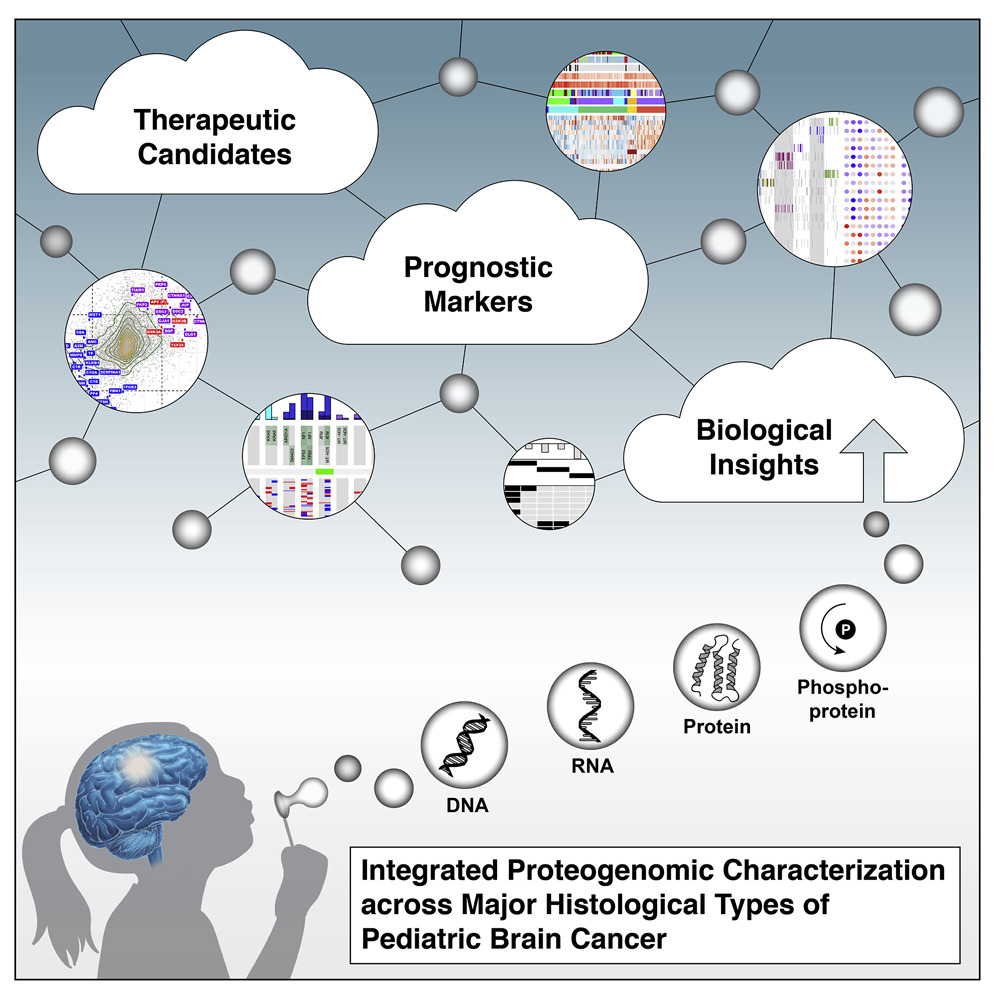
Brains for the Cure’s writers review several important developments in recent pediatric brain tumor research.
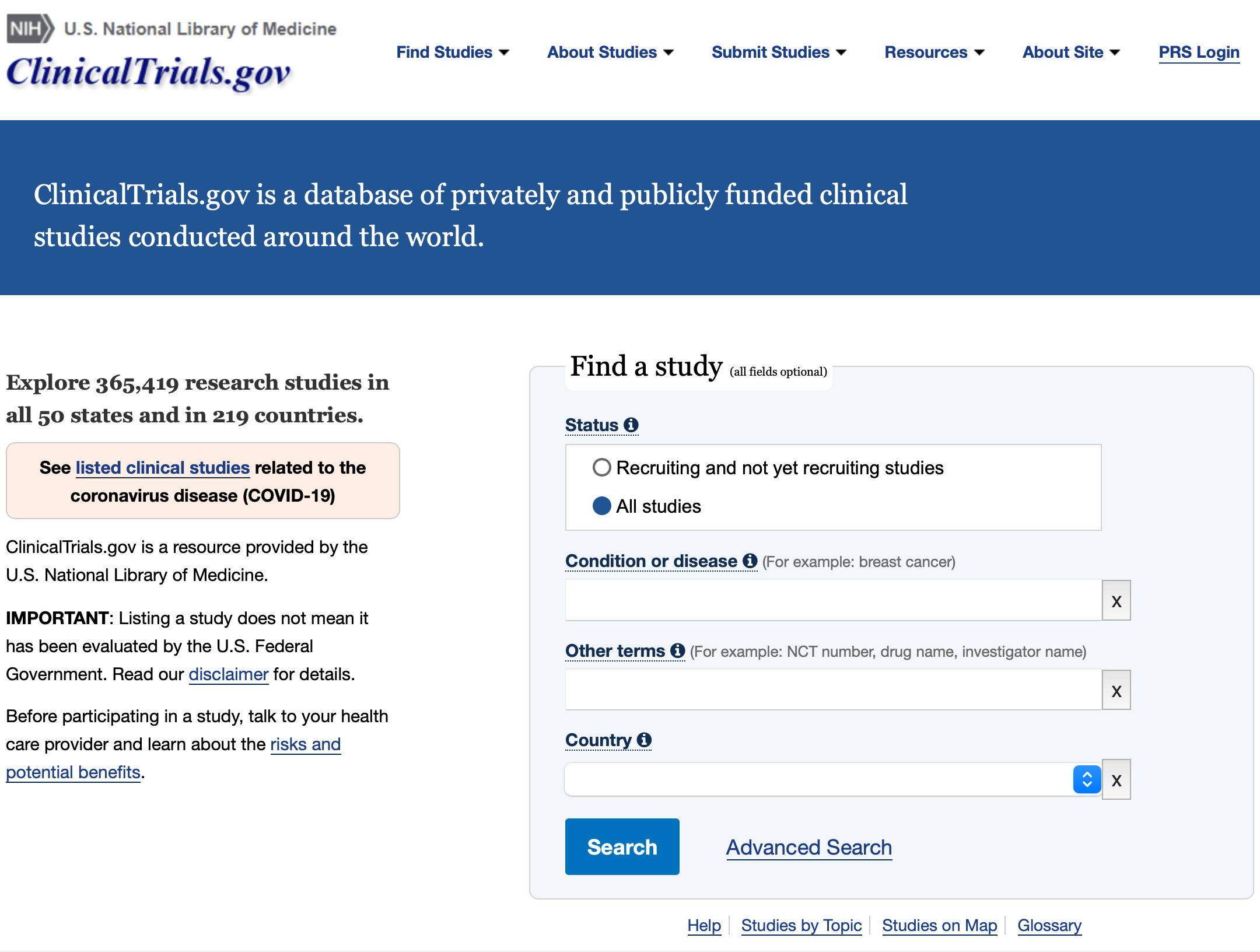
The website ClinicalTrials.gov is one that that patients and families fighting brain tumors should be familiar with. Knowing how to use the site yourself or