The website ClinicalTrials.gov is one that that patients and families fighting brain tumors should be familiar with. Knowing how to use the site yourself or with the help of staff from the cancer center where they receive care can help you make more informed treatment decisions.
Why is this particular government-run website important? Because clinical trials are needed for better treatment of brain tumors. Aggressive brain tumors remain one of the hardest forms of cancer to treat. The gold standard of medical research is clinical trials, which are really a kind of collaboration between willing patients and clinicians exploring a new treatment approach.
Not only are they the key to medical advancement, they are a way for patients to receive cutting-edge care that could make a difference in their survival.

Why the government provides ClinicalTrials.gov
At the same time, matching patients and suitable trials is a longstanding challenge. Families try and access a trial, only to hear that they don’t meet eligibility requirements, due to factors they cannot control, such as the genetics of their tumor or perhaps a treatment decision they made earlier in their journey, often in emergency circumstances. And while these requirements are necessary for researchers to do rigorous science, they also create situations where discovery moves forward slowly or not at all, because trials cannot recruit the minimum number of participants.
ClinicalTrials.gov is a website run by the National Institutes of Health that publishes information about clinical trials in the United States that are currently seeking patients or in progress. The site is required by law to publish information about all studies that receive federal funding and, as of 2002, most that receive private funding.
And as a public resource, the site will never try and misuse your private information, display advertising, or display commercial content.
Effectively Using ClinicalTrials.gov
The people conducting a trial, often referred to as “investigators,” are always medical doctors and trained scientists who work within a strict framework of patient safety and transparency, which is upheld by the Food and Drug Administration, the NIH, and the institutions that employ them (typically research medical centers associated with a major university).
The site allows investigators to enter the details and requirements of the clinical trials that they are conducting. Then, patients and other doctors can use the site to search these details and find trials that are a match for their circumstances.
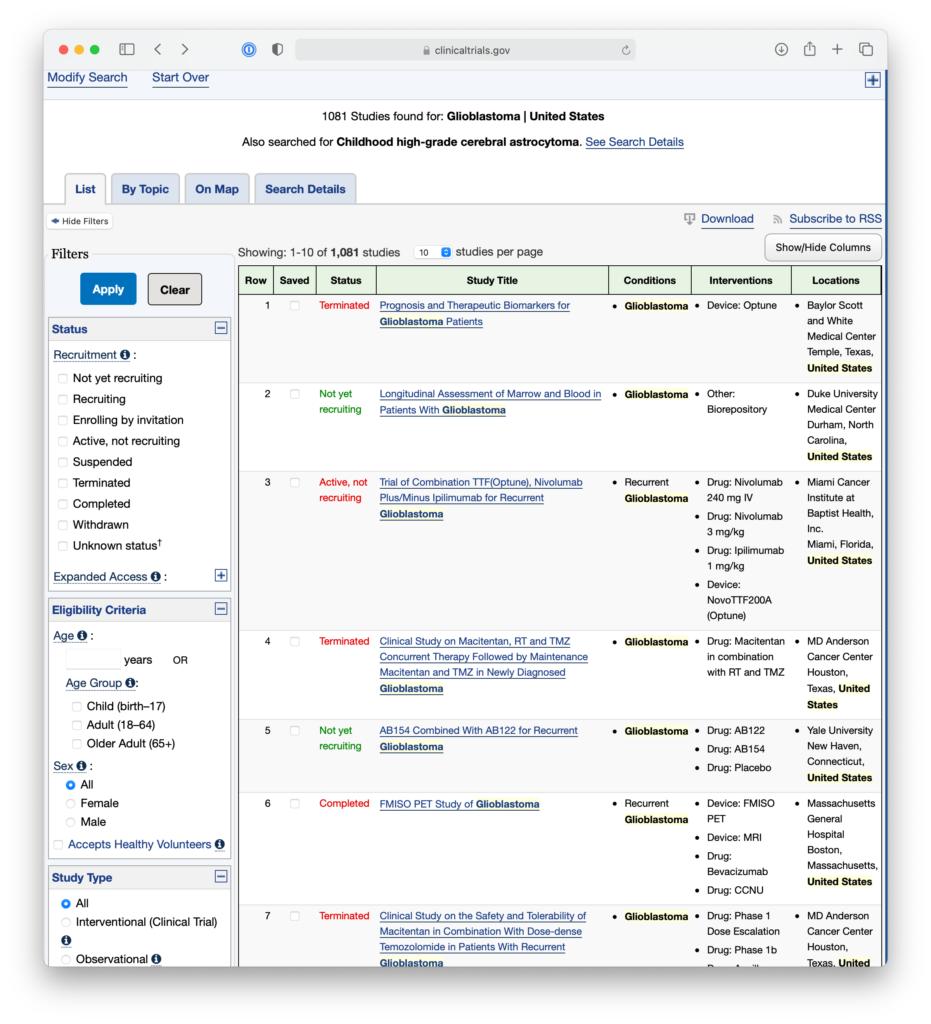
For example, patients might enter Glioblastoma in the search field, set the country to United States, and then leave the other location fields blank in order to search for trials across the United States. They’re then taken to a results page, where they may see a number of trials that are concluded, or no longer recruiting, or seeking patients outside their age group.
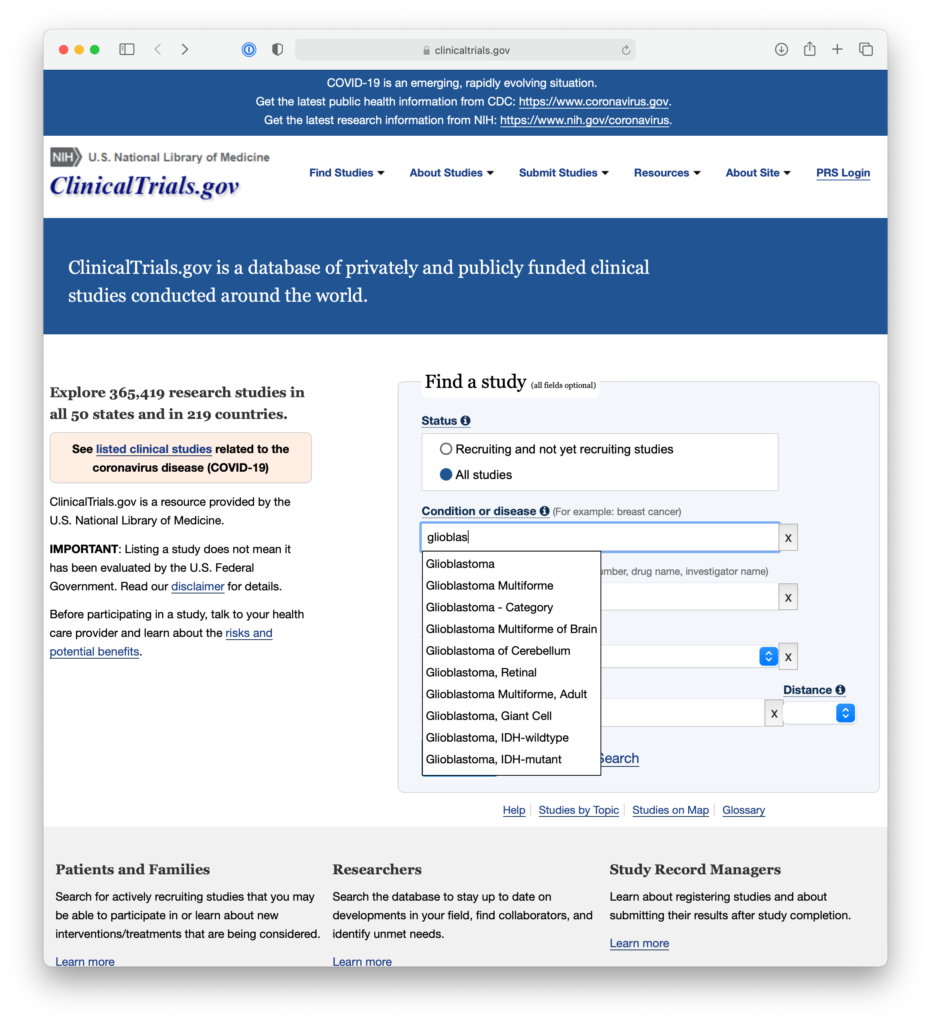
At this time, they could use the filters on the right to show only trials that are currently recruiting adult patients, so they could get a list of trials that they might be able to inquire further about.
If they click on a study, they are taken to a page called a Study Record. These pages contain eligibility requirements and contact information for investigators, so interested patients or doctors can reach out for more information.
These pages often contain medical language and specialized terminology, especially when describing the granular details of eligibility. Therefore, even if a patient is conducting an initial search for trials themselves, at this point it would be useful for them to discuss trials they’re found with doctors or nurses who specialize in brain tumor treatment.
ClinicalTrials.gov is a powerful resource, because it gives patients some ability to find options beyond what their existing team tells them about. It cannot substitute for reaching out and talking to doctors and teams, but it can be a useful first step.
Often, trial eligibility is a complex, so it’s important not to get discouraged if you reach out about a trial and are told you are not eligible.
And if you already have your team in place, you can use the site alongside your team to find options and approaches you may have missed.
Related resources
- ClinicalTrials.gov – homepage where you access the main search tool
- How To Use Search – an informative page about effectively searching clinicaltrials.gov
- How to Read a Study Record
- NCI Steps to Find a Clinical Trial – a step-by-step guide from the National Cancer Institute of the NIH about finding trials
- NCI-supported Clinical Trials – a search tool similar to ClinicalTrials.gov, with simpler search and results (but contains fewer trials)
- Questions To Ask About Trials – a list of resources from Brains for the Cure to better understand clinical trials and make informed decisions.


