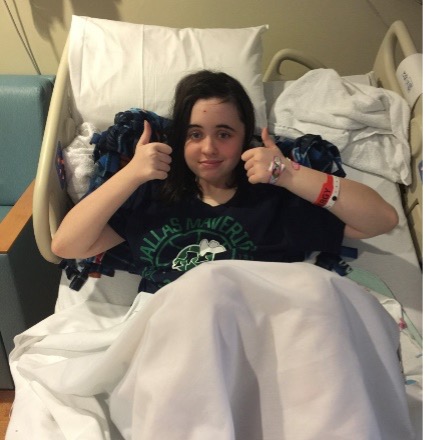
A Childhood Interrupted by Brain Cancer
Anna right after diagnosis at age 12. Anna’s first HFTC 5K run/walk at age 14. Another 5K run/walk at age 15. Graduating high school at
Brains for the Cure - Brain Tumor News
Get Help With Brain Tumors

Anna right after diagnosis at age 12. Anna’s first HFTC 5K run/walk at age 14. Another 5K run/walk at age 15. Graduating high school at

Written by Optune® Ambassador Lynn Lynn was living a fast-paced life before she was diagnosed with glioblastoma (GBM). Since her diagnosis, she’s shifted her perspective and

Head for the Cure Ambassador and Survivor, Kyla Burridge shares why she chose to participate in a clinical trial, and why trials are vital to finding a cure for brain tumors.

Written by Optune® Ambassador Ashley M.J. Hello everyone, my name is Ashley. If I had to describe myself in one word, it would be “grateful”.

Written by Optune® Ambassador Gill D. Hi, everyone, my name is Gill. I’d like to take this opportunity to thank Novocure for making it possible

Written by Optune® Ambassador Eric C. My father used to say to me when I was a kid, “Eric, don’t be helpless.” Throughout my life

Written by Optune Ambassador Tim H. I think probably the most important thing to know about me is that I’m always looking for the good