Clinical Trials
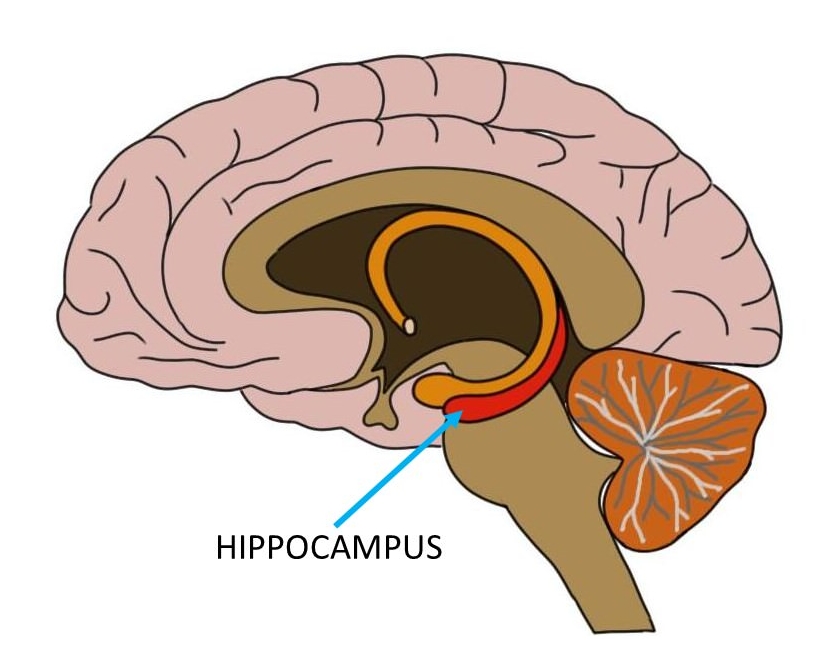
Preserving Cognitive Function for Patients with Brain Metastases
At the 24th Annual Meeting and Education Day of the Society for NeuroOncology (SNO), there were hundreds of presentations and poster sessions detailing the latest
Brains for the Cure - Brain Tumor News
Get Help With Brain Tumors